How are newly synthesized proteins inserted in the membrane?
Mechanism varies with the type of protein
Type I: Signal sequence on amino terminus enters first and continues to elongate. Protein is threaded through the translocating channel (open area in rer membrane) until a hydrophobic stop sequence is reached. That hydrophobic stop sequence (seen as a hatched region in the protein) is then inserted in the membrane and forms the anchor for that protein. Signal is cleaved by protease inside the lumen. Type II: No cleavable signal sequence. These proteins have rather long hydrophobic regions that will be anchored in the membrane. Type II proteins are threaded into the lumen with the C terminus leading. Protein continues to be inserted until it reaches the hydrophobic stop signal sequence. Type III: Same as Type II, only the N terminus leads into the lumen.
What regulates the orientation of Type II and III proteins?
Wahlberg, JM. And Spiess, M. Multiple determinants direct the orientation of signal anchor proteins: The topogenic role of the hydrophobic signal domain. J Cell Biology 137: 555-562.
Tested charged amino acids and length of hydrophobic signal sequence. The "positive inside rule" states that amino acid residues nearest the cytosolic side of the hydrophobic anchor sequence are more positive than those nearest the lumenal side. So, whichever end has the least positive charges near the signal anchor patch would go into the ER lumen. One can change the direction of translocation of a protein (reverse it) by mutating the protein and making more positively charged groups near the anchor patch of the other end. Below, the cartoon shows that this can be done to change a Type II protein (COOH end enters ER lumen) to a Type III (which has its amino terminal entering the lumen).Washburn and Speiss (JCB 137: 555-562, 1997) also tested the length of the hydrophobic signal anchor sequence. The following cartoon shows that a longer hydrophobic anchor sequence (seen as the portion running through the membrane) promotes entry with the amino terminal leading into the lumen.
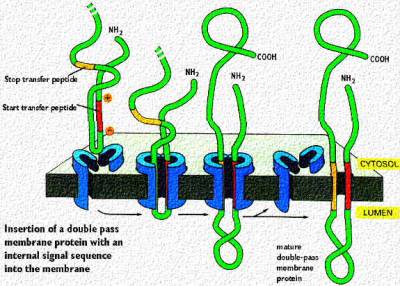
Proteins destined for insertion into membranes, such as ion channels or receptors have mRNA codes for start and stop sequences that allow multiple passes through the membrane. Signalling sequences (patches) can be formed as described in the above cartoon. It shows the insertion of a double pass transmembrane protein with the loop inside the rough endoplasmic reticulum. The red signal patch has the + charges near the cytosol and starts the insertion process. This continues until the hydrophobic stop signal patch is reached. That anchors the second membrane passage. Note, that both C and N terminal portions are in the cytosol Thus, if this protein is destined for the plasma membrane, that loop in the ER lumen will eventually project outside the cell Membrane proteins that pass through the membrane multiple times (called "multipass transmembrane proteins) have multiple start and stop signals. They are aligned with the hydrophilic and hydrophobic portions of the lipid bilayer as described in the lecture on membranes. This is shown in the following cartoon
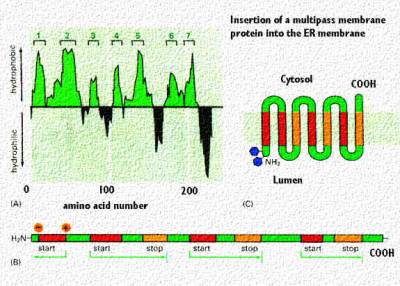
|




